   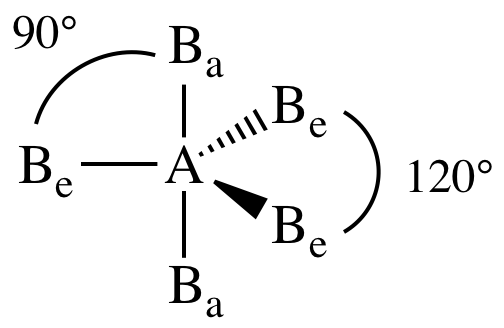
Sulfur tetrafluoride is the premier example of a molecule with the disphenoidal molecular geometry (see image at upper right). The four atoms in motion act as a lever about the central atom for example, the four fluorine atoms of sulfur tetrafluoride rotate around the sulfur atom. The F-S-F bond angles in the SF 4 molecule are 101.6° and 187° respectively. The central S atom in SF 4 is sp 3 d hybridized. Seesaw molecular geometry VSEPR theory Trigonal bipyramidal molecular geometry, others, chemistry, seesaw Molecular Geometry, square Planar Molecular Geometry. In the Lewis dot structure of SF 4, there are a total of 4 bond pairs and 1 lone pair around the central sulfur atom. A sp3d hybridized has a molecular geometry of seesaw, how many lone pairs are. The three bond angles in the middle (equatorial position) are 120 degrees apart and the two electron groups in the axial positions on opposite sides of the equatorial plane are 90 degrees from the plane. Thus, the 19F NMR spectrum of SF 4 (like that of PF 5) consists of single resonance near room temperature. The electron geometry of SF 4 is trigonal bipyramidal. Explain the bond angles in trigonal bipyramidal geometry. you can identify it on a VSEPR chart as a molecule with a trigonal bipyramidal molecular geometry. The angle between the axial ligands and the equatorial ligands is 90° whereas the angle between the two equatorial ligands themselves is 120°.ĭisphenoidal molecules, like trigonal bipyramidal ones, are subject to Berry pseudorotation. The structures are: linear, trigonal planar, angled, tetrahedral, trigonal pyramidal, trigonal bipyramidal, disphenoidal (seesaw), t-shaped, octahedral, square pyramidal, square planar, and pentagonal bipyramidal. Typically the bond distance to the apical ligands is longer than to the equatorial ligands. A separate pair of ligands is situated orthogonal to the axial ligands. Compounds with disphenoidal geometry (See-Saw Geometry) have two types of ligands, one pair related by 180° often called axial ligands. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
